Recently, Prof. Dehui Deng and Assoc. Prof. Liang Yu from Group 05T6 in State Key Laboratory of Catalysis (SKLC) of Dalian Institute of Chemical Physics (DICP) made a new process in the field of methane conversion to oxygenates under mild conditions. They reported that highly-dispersed coordinatively unsaturated Fe-sites confined within the ZSM-5 channels enable highly efficient methane conversion to formic acid (HCOOH) under mild conditions.
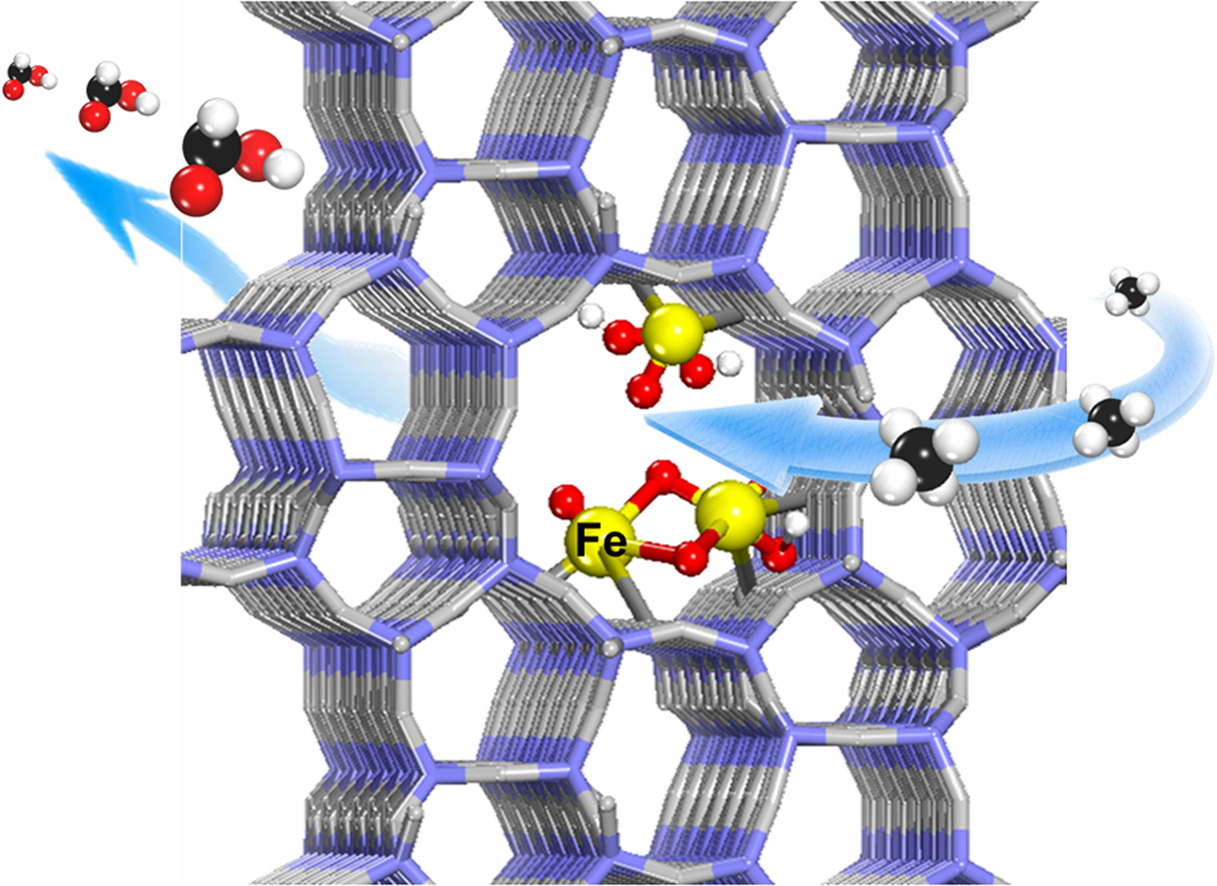
Methane conversion to formic acid on the atomically-dispersed Fe sites confined within the channels of ZSM-5
Methane, as the main component of shale gas, natural gas and combustible ice, is among the most promising energy resources for producing high-value-added chemicals. Methane conversion to value-added chemicals or fuels under mild conditions has become one of the hottest topics in energy and catalysis with the need of developing more economical and environmental-friendly path in contrast to the energy-intensive industrial process. However, the high symmetry and low polarizability of methane molecule render it highly challenging to activate methane under mild conditions. In addition, the target products (e.g., C1 oxygenates) are usually more reactive than the methane, which are subject to over-oxidation to the greenhouse gas CO2. Hence, selective activation and directional conversion of methane to high-value-added chemicals or fuels under mild conditions is of great significance in catalysis, which requires innovative modulation for the balance between catalytic activity and selectivity.
Based on their previous study on the C-H activation and methane conversion to C1 oxygenates at room temperature (Sci. Adv. 2015, 1, e1500462; Nat. Commun. 2018, 9, 1181; Chem 2018, 4, 1902), Prof. Deng’s research teamreported that highly efficient and selective oxidation of methane to HCOOH can be achieved on the atomically-dispersed Fe sites confined within the channels of ZSM-5. Via adjusting the silica-alumina ratio and Fe loading in the ZSM-5 to modulate the microenvironment of the active Fe species, a turnover frequency (TOF) of 84200 h-1 for producing C1 oxygenates and a high HCOOH selectivity of 91% with productivity of 383.2 mmol gcat.-1 h-1 are reached at 80 oC, which surpass all previously reported methane conversion catalysts operated under similar conditions. Combining various characterizations and density functional theory calculations, they found that Fe-O active sites can be generated from H2O2 dissociation on both the mononuclear and binuclear Fe centers confined within the channels of ZSM-5. The Fe-O active sites facilitate activation and cleavage of the C-H bond of methane and thereby promote successive oxidation of methane to formic acid through methanol and formaldehyde via free radical pathways, while inhibiting the over-oxidation to CO2. This study paves a new route to design and construct lattice-confined active sites for highly efficient conversion of methane under mild conditions.
This work has been published as a research article in Nano Energy. This work was supported by the National Key R&D Program of China, the National Natural Science Foundation of China, the Strategic Priority Research Program of Chinese Academy of Sciences, the DNL Cooperation Fund, CAS, etc. (Text by Kaixin Zhu and Hehua Gao)
Link: http://www.dicp.ac.cn/xwdt/kyjz/202101/t20210104_5849986.html